The International Tinnitus Journal
Official Journal of the Neurootological and Equilibriometric Society
Official Journal of the Brazil Federal District Otorhinolaryngologist Society
ISSN: 0946-5448

Google scholar citation report
Citations : 12717
The International Tinnitus Journal received 12717 citations as per google scholar report
The International Tinnitus Journal peer review process verified at publons
Indexed In
- Excerpta Medica
- Scimago
- SCOPUS
- Publons
- EMBASE
- Google Scholar
- Euro Pub
- CAS Source Index (CASSI)
- Index Medicus
- Medline
- PubMed
- UGC
- EBSCO
Volume 21, Issue 2 / December 2017
Case Report Pages:128-132
Vestibular Schwannoma and Ipsilateral Endolymphatic Hydrops: An Unusual Association
Authors: Massimo Ralli, Antonio Greco, Giancarlo Altissimi, Rosaria Turchetta, Lucia Longo, Vittorio D’Aguanno, Mario Tombolini, Giancarlo Cianfrone, Marco de Vincentiis
PDF
Abstract
Vestibular schwannoma and endolymphatic hydrops are two conditions that may present with similar audio-vestibular symptoms. The association of the two in the same patient represents an unusual finding that may lead clinicians to errors and delays in diagnosis and clinical management of affected subjects. We discuss the case of a patient with an intrameatal vestibular schwannoma reporting symptoms suggestive for ipsilateral endolymphatic hydrops. The patient presented with fluctuating hearing loss, tinnitus, and acute rotatory vertigo episodes, and underwent a full audiological evaluation and imaging of the brain with contrast-enhanced Magnetic Resonance Imaging. Clinical audio-vestibular and radiological examination confirmed the presence of coexisting vestibular schwannoma and endolymphatic hydrops. Hydrops was treated pharmacologically; vestibular schwannoma was monitored over time with a wait and scan protocol through conventional MRI. The association of vestibular schwannoma and endolymphatic hydrops is rare, but represents a possible finding in clinical practice. It is therefore recommended investigating the presence of inner ear disorders in patients with vestibular schwannoma and, similarly, to exclude the presence of this condition in patients with symptoms typical of inner ear disorders.
Keywords: vestibular schwannoma, endolymphatic hydrops, hearing loss, tinnitus
Keywords
vestibular schwannoma, endolymphatic hydrops, hearing loss, tinnitus
Introduction
Vestibular schwannomas (VS), also known as acoustic neuromas, are benign neoplasms originating from the Schwann cells of the eighth cranial nerve and present most frequently in subjects between 30 and 60 years [1]. VS have a prevalence of 1.4-2.1 in 10.000 people [2,3], and represent 5% to 10% of all intracranial tumors, and 80% to 90% of neoplasms of the cerebellopontine angle [4].
VS presenting symptoms usually include slowly progressive sensorineural hearing loss, fullness and tinnitus associated to vestibular disorders in 16-70% of cases; balance disorders can present with acute rotatory vertigo and/or dizziness [5,6]. Medical history, clinical examination and routine audio-vestibular examination alone cannot diagnose VS; diagnosis must be confirmed by high-resolution Magnetic Resonance Imaging (MRI) with intravenous gadolinium-based contrast [7]. Along with imaging technology improvements, the number of VS diagnosis has increased including smaller schwannomas previously overlooked [8].
The presenting symptoms of VS can also be found in other inner ear disorders such as endolymphatic hydrops (ELH); nevertheless, inner ear disorders may be present in patients with VS [9]. ELH is a disorder of the inner ear characterized by episodic attacks of vertigo, tinnitus, fluctuating hearing loss, ear fullness, and progressive loss of the audio-vestibular function [10].
Differential diagnosis plays a central role for a correct diagnostic and therapeutic management of VS patients; at the same time, it is important not to overlook other concomitant inner ear disorders [10].
In this paper, we describe the case of a patient with fluctuating hearing loss, tinnitus and acute rotatory vertigo episodes, who presented an intrameatal vestibular schwannoma and a symptomatology suggestive for ipsilateral endolymphatic hydrops.
Case Presentation
A 67-year-old man was admitted to the Otolaryngology Department of the Policlinico Umberto I, Sapienza University of Rome in December 2013 with a three-year history of fluctuating hearing loss in the left ear and vertigo attacks lasting 2 to 6 hours associated to intense nausea and vomiting (4-6 episodes/year). Last episode occurred one month before admission.
After admission, patient underwent a complete otolaryngology examination with otoscopy, Pure-Tone Audiometry (PTA), Auditory Brainstem Response (ABR), Distorsion-Product Otoacoustic Emissions (DPOAE), Infrared Videonystagmography (IR-VNG) for spontaneous and gaze-evoked nystagmus and Fitzgerald-Hallpike caloric stimulation. Vestibular examination was completed with Cervical Vestibular Evoked Myogenic Potential (c-VEMP) and Ocular Vestibular Evoked Myogenic Potential (o-VEMP) response recording. Written informed consent was obtained from the patient for all the diagnostic and therapeutic procedures performed.
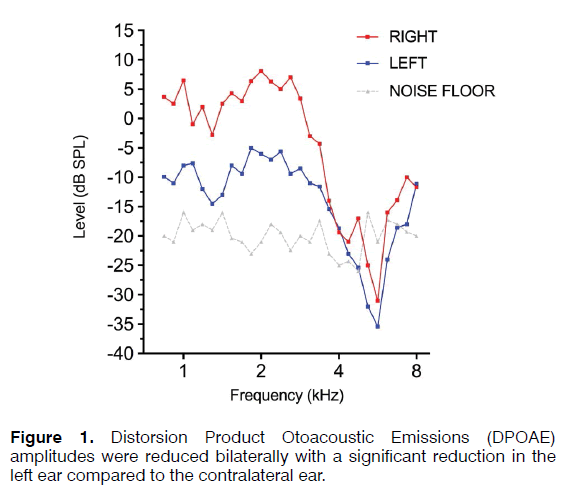
The patient had a normal otoscopy; PTA showed a mild sensorineural hearing loss in the right ear with an average auditory threshold of 37.50 dB and a moderate sensorineural hearing loss in the left ear with an average threshold of 61.25 dB, more evident for mid-low frequencies. Weber test lateralized to the right at all frequencies. Speech Discrimination was in accordance with PTA; no alterations were found at ABR (Table 1); DPOAE amplitudes were reduced bilaterally with a significant reduction in the left ear compared to the contralateral ear (Figure 1). No spontaneous or gazeevoked nystagmus was found at IR-VNG; left hyporeflexia was observed at the caloric test. The first c-VEMP complex of biphasic wave p13-n23 and the n10 response of the o-VEMP to Bone Conducted Vibration and Air Conducted Sound were normally evoked bilaterally.
| I | III | V | I-III | III-V | I-V | |
| Right | 1.58 | 3.72 | 5.68 | 2.13 | 1.97 | 4.10 |
| Left | 1.68 | 3.81 | 5.63 | 2.12 | 1.82 | 3.95 |
Table 1. Auditory Brainstem Response recording in our patient. No alterations can be seen for waveforms and latencies in both sides.
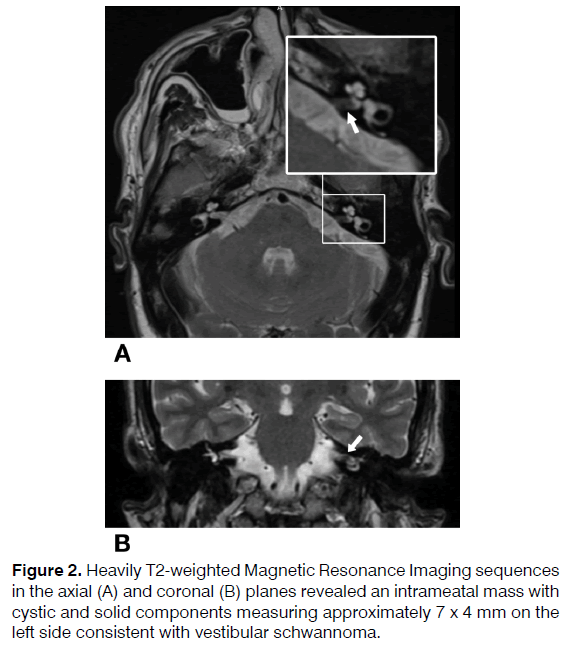
Audiological examination was followed by a conventional MRI of the brain with gadolinium-based contrast agent performed in the axial, sagittal and coronal planes focusing on the eight cranial nerve and cerebellopontine angle; heavily T2-weighted sequences revealed an intrameatal mass with cystic and solid components measuring approximately 7 x 4 mm on the left side consistent with vestibular schwannoma (Figure 2).
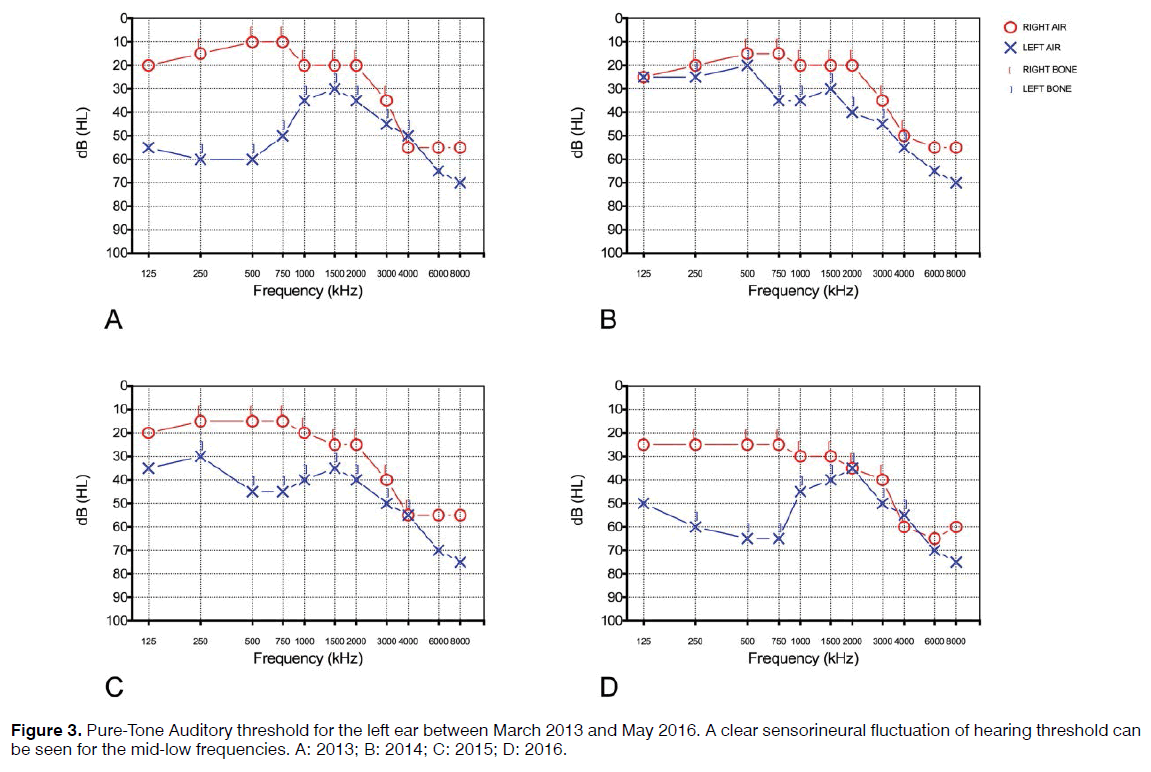
During the following 30 months, the patient experienced fluctuating hearing loss in the left ear associated to acute vertigo attacks monitored over time with PTA and IR-VNG; exams documented a fluctuation of hearing threshold in the mid-low frequency range in the left ear with average auditory threshold for frequencies 500-4000 Hz of 53.75 dB in March 2013, 32.5 dB in February 2014, 45.75 in March 2015 and 61.25 in May 2016 (Figure 3). When recorded during the vertigo attack, IR-VNG showed a spontaneous left beating nystagmus. The patient also reported the onset of a stable high-pitch tinnitus in the left ear one year after initial admission to our unit.
The results of the audiological evaluation during follow up led towards a diagnosis of left VS associated to an ipsilateral ELH. Diagnosis of ELH was posed according to AAO-HNS Committee on Hearing and Equilibrium classification criteria [11,12] but was not confirmed radiologically due to unavailability of 3-Tesla MRI. Hydrops was treated pharmacologically with betahistine-dihydrochloride during crisis and long-term cinnarizine plus chlorthalidone associated to low sodium diet following our internal management protocol; VS was monitored over time with a wait and scan protocol through conventional MRI.
Discussion
The presentation of simultaneous VS and ELH on the same side in the same patient is uncommon and has seldom been described in the literature [13-16]. Although the two pathologies are most probably of independent origin and should be considered separate entities, presenting and ongoing symptoms may overlap and may cause an incorrect treatment of the cochlear disorder (i.e. misdiagnosis of the endolymphatic hydrops), a potential delay in VS diagnosis (i.e. postponing the execution of a MRI) or a potential misinterpretation of VS evolution (i.e. attributing symptoms to VS growth instead of inner ear dysfunction, or vice-versa) [13,16-19].
In such cases, it is always puzzling to attribute the vestibular symptoms to the inner ear or to the retrocochlear condition. Several studies discussed the origin (retrocochlear or cochlear) of audio-vestibular symptoms in patients with VS, suggesting that vertigo and hearing loss may also have a peripheral origin, possibly induced by tumor-related vascular alterations of the inner ear, rather than being exclusively linked to a direct pressure of the tumor on the vestibulocochlear nerves [20,21]. Roosli et al studied temporal bones from 32 patients with unilateral VS, finding significantly more hair cell loss, cochlear neuronal loss and decreased pure tone average when compared with the opposite ear; however, tumor size, distance from the cochlea, and nerve of origin did not correlate with structural changes found in the cochlea [20].
Due to the varying and fluctuating symptoms, the clinical diagnosis of ELH may be difficult [22]. The role of new imaging techniques is gaining importance in ELH diagnosis, since conventional MRI is unable to distinguish the endolymphatic from the perilymphatic compartments within the inner ear [23]. In the recent years, several efforts have been made to develop reliable imaging able to identify ELH; some inner ear imaging techniques enable visualizing morphological changes in the cochlea and the vestibulum in vivo [24]. Current research demonstrates the validity of Locally Enhanced Inner Ear MRI (LEIM) after intratympanic contrast agent application and 3-Tesla MRI performed with intravenous gadolinium with 3D-FLAIR sequences for the diagnosis and grading of ELH; these techniques have shown similar sensitivity to histopathological series in identifying ELH [25-27]. The degree of perilymphatic enhancement in 3-Tesla MRI also depend on the interval between the intravenous contrast administration and imaging. Research showed that a 4-hour delay results in greater contrast enhancement within the perilymph [28,29]. Jerin et al reported a case of a patient with VS and endolymphatic hydrops, in which ELH was revealed with the use of an inner ear LEIM MRI with an axial 3D Real-IR sequence; the authors recommended performing locally contrast-enhanced inner ear MRI in those cases in which an ELH is suspected [14]. In a study from Kentala and Pyykkö, the authors retrieved information on 127 patients with VS and 243 patients with ELH and found that 38% of the VS patients had the full triad of vertigo, hearing loss and tinnitus and vertigo mimicked that in ELH in 14% of cases [19]. In a recent study to verify whether vertigo in patients with VS correlates to vestibular endolymphatic hydrops, Naganawa et al studied 15 patients with unilateral VS using a noncontrast- enhanced 3D FLAIR MRI and reported vestibular hydrops in 4/15 cases; however, no significant correlation between vertigo and hydrops was found [15].
In our patient, the two pathological conditions coexist but are most probably unrelated. The full audiological and radiological protocol performed allowed to identify separately both cochlear and retrocochlear disorders, although the diagnosis of ELH was not confirmed by imaging due to unavailability of 3-Tesla MRI (the availability of 3-Tesla scanners is still limited in many hospitals) or, as suggested by some authors, electrocochleography [30]; consequently, ELH diagnosis was only based on symptoms reported by the patient over the years associated to the results of the auditory and vestibular evaluation performed initially and during follow up and to patient’s response to therapy.
Conclusion
Although current literature supports the notion of a peripheral origin of vertigo in patients with small vestibular schwannomas, it is always recommended in patients with VS to also investigate possible coexisting independent inner ear disorders such as ELH, especially when a fluctuation in audio-vestibular symptoms is documented. When available, a 3-Tesla MRI performed at 4 hours post-intravenous gadolinium with 3D-FLAIR sequences or a locally contrast-enhanced inner ear MRI to detect endolymphatic hydrops may be useful to confirm ELH. At the same time, as widely documented in the literature, it is always recommended performing gadolinium contrastenhanced MRI in patients with unilateral hearing loss and symptoms suggesting a peripheral origin to exclude retrocochlear lesions such as VS. Unfortunately, the need for a thorough neuro-otological work-up including MRI scanning can still be overlooked in the cases in which it appears to be a clear peripheral etiology for the hearing loss such as in endolymphatic hydrops.
Conflicts of Interest
None
Source of Funding
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
References
- Stangerup SE, Caye-Thomasen P. Epidemiology and natural history of vestibular schwannomas. Otolaryngol Clin North Am. 2012;45(2):257-68.
- Propp JM, McCarthy BJ, Davis FG, Preston-Martin S. Descriptive epidemiology of vestibular schwannomas. Neuro Oncol. 2006;8(1):1-11.
- Lin D, Hegarty JL, Fischbein NJ, Jackler RK. The prevalence of "incidental" acoustic neuroma. Arch Otolaryngol Head Neck Surg. 2005;131(3):241-4.
- Hart RG, Davenport J. Diagnosis of acoustic neuroma. Neurosurgery. 1981;9(4):450-63.
- Espinosa-Sanchez JM, Lopez-Escamez JA. Meniere's disease. Handb Clin Neurol. 2016;137:257-77.
- Gurkov R, Pyyko I, Zou J, Kentala E. What is Meniere's disease? A contemporary re-evaluation of endolymphatic hydrops. J Neurol. 2016;263(Suppl 1):S71-81.
- Coffey N, Torres C, Glikstein R, Al Mansoori T, Del Carpio-O'Donovan R, Patro S. Imaging findings in sensorineural hearing loss: a pictorial essay. Can Assoc Radiol J. 2017;68(2):106-15.
- Carlson ML, Link MJ, Wanna GB, Driscoll CL. Management of sporadic vestibular schwannoma. Otolaryngol Clin North Am. 2015;48(3):407-22.
- Ralli M, Nola G, Sparvoli L, Ralli G. Unilateral enlarged vestibular aqueduct syndrome and bilateral endolymphatic hydrops. Case Rep Otolaryngol. 2017;2017:6195317.
- Slattery EL, Babu SC, Chole RA, Zappia JJ. Intralabyrinthine schwannomas mimic cochleovestibular disease: Symptoms from tumor mass effect in the labyrinth. Otol Neurotol. 2015;36(1):167-71.
- Lopez-Escamez JA, Carey J, Chung WH, Goebel JA, Magnusson M, Mandala M, et al. Diagnostic criteria for Meniere's disease. J Vestib Res. 2015;25(1):1-7.
- Committee on Hearing and Equilibrium. Report of subcommittee on equilibrium and its measurement. Meniere's disease: Criteria for diagnosis and evaluation of therapy for reporting. Trans Am Acad Ophthalmol Otolaryngol. 1972;76(6):1462-4.
- Morrison GA, Sterkers JM. Unusual presentations of acoustic tumours. Clin Otolaryngol Allied Sci. 1996;21(1):80-3.
- Jerin CK, E; Ertl-Wagner, B; Gürkov, R. Endolymphatic hydrops in a patient with a small vestibular schwannoma suggests a peripheral origin of vertigo. Austin J Radiol. 2015;2(6):1033.
- Naganawa S, Kawai H, Sone M, Nakashima T, Ikeda M. Endolympathic hydrops in patients with vestibular schwannoma: Visualization by non-contrast-enhanced 3D FLAIR. Neuroradiology. 2011;53(12):1009-15.
- Dispenza F, De Stefano A, Flanagan S, Romano G, Sanna M. Decision making for solitary vestibular schwannoma and contralateral Meniere's disease. Audiol Neurootol. 2008;13(1):53-7.
- You H, Li X, Wang W. [Vestibular schwannoma: A case report of misdiagnosis]. JAMA Otolaryngol Head Neck Surg. 2014;28(22):1809-11.
- Weber PC, Adkins WY Jr. The differential diagnosis of Meniere's disease. Otolaryngol Clin North Am. 1997;30(6):977-86.
- Kentala E, Pyykko I. Vestibular schwannoma mimicking Meniere's disease. Acta Otolaryngol Suppl. 2000;543:17-9.
- Roosli C, Linthicum FH Jr, Cureoglu S, Merchant SN. Dysfunction of the cochlea contributing to hearing loss in acoustic neuromas: An underappreciated entity. Otol Neurotol. 2012;33(3):473-80.
- Gouveris HT, Victor A, Mann WJ. Cochlear origin of early hearing loss in vestibular schwannoma. Laryngoscope. 2007;117(4):680-3.
- Lingam RK, Connor SEJ, Casselman JW, Beale T. MRI in otology: Applications in cholesteatoma and Meniere's disease. Clin Radiol. 2017.
- Tanioka H, Kaga H, Zusho H, Araki T, Sasaki Y. MR of the endolymphatic duct and sac: Findings in Meniere disease. AJNR Am J Neuroradiol. 1997;18(1):45-51.
- Gurkov R, Berman A, Dietrich O, Flatz W, Jerin C, Krause E, et al. MR volumetric assessment of endolymphatic hydrops. Eur Radiol. 2015;25(2):585-95.
- Sepahdari AR, Ishiyama G, Vorasubin N, Peng KA, Linetsky M, Ishiyama A. Delayed intravenous contrast-enhanced 3D FLAIR MRI in Meniere's disease: Correlation of quantitative measures of endolymphatic hydrops with hearing. Clin Imaging. 2015;39(1):26-31.
- Tagaya M, Teranishi M, Naganawa S, Iwata T, Yoshida T, Otake H, et al. 3 Tesla magnetic resonance imaging obtained 4 hours after intravenous gadolinium injection in patients with sudden deafness. Acta Otolaryngol. 2010;130(6):665-9.
- Pyykko I, Nakashima T, Yoshida T, Zou J, Naganawa S. Meniere's disease: A reappraisal supported by a variable latency of symptoms and the MRI visualisation of endolymphatic hydrops. BMJ Open. 2013;3(2).
- Kim TY, Park DW, Lee YJ, Lee JY, Lee SH, Chung JH, et al. Comparison of Inner ear contrast enhancement among patients with unilateral inner ear symptoms in MR images obtained 10 minutes and 4 hours after gadolinium injection. AJNR Am J Neuroradiol. 2015;36(12):2367-72.
- Sano R, Teranishi M, Yamazaki M, Isoda H, Naganawa S, Sone M, et al. Contrast enhancement of the inner ear in magnetic resonance images taken at 10 minutes or 4 hours after intravenous gadolinium injection. Acta Otolaryngol. 2012;132(3):241-6.
- Ferraro JA, Durrant JD. Electrocochleography in the evaluation of patients with Meniere's disease/endolymphatic hydrops. J Am Acad Audiol. 2006;17(1):45-68.
References
1Department of Oral and Maxillofacial Sciences, Sapienza University of Rome, Italy
2Department of Sense Organs, Sapienza University of Rome, Italy
Send correspondence to:
Massimo Ralli
Department of Oral and Maxillofacial Sciences, Sapienza University of Rome. Viale del Policlinico 155, 00186, Rome Italy, E-mail: massimo.ralli@uniroma1.it
Paper submitted to the ITJ-EM (Editorial Manager System) on October 18, 2017; and accepted on October 31, 2017.
Citation: Ralli M, Greco A, Altissimi G, Turchetta R, Longo L, D’Aguanno V, Tombolini M, Cianfrone G, Vincentiis MD. Vestibular Schwannoma and Ipsilateral Endolymphatic Hydrops: An Unusual Association. Int Tinnitus J. 2017;21(2):128-132