The International Tinnitus Journal
Official Journal of the Neurootological and Equilibriometric Society
Official Journal of the Brazil Federal District Otorhinolaryngologist Society
ISSN: 0946-5448

Google scholar citation report
Citations : 12717
The International Tinnitus Journal received 12717 citations as per google scholar report
The International Tinnitus Journal peer review process verified at publons
Indexed In
- Excerpta Medica
- Scimago
- SCOPUS
- Publons
- EMBASE
- Google Scholar
- Euro Pub
- CAS Source Index (CASSI)
- Index Medicus
- Medline
- PubMed
- UGC
- EBSCO
Volume 27, Issue 1 / June 2023
Research Article Pages:35-39
10.5935/0946-5448.20230006
Factors Affecting Early and Late Cochlear Implantation
Authors: Wan Nabila Wan Mansor, Asma Abdullah, Goh Bee See, Cila Umat, Shamsul Azhar Shah
PDF
Abstract
Objectives: This study aimed to describe the factors affecting early and late cochlear implantation.
Materials and Methods: A total of 159 patients from the Universiti Kebangsaan Malaysia (UKM) Cochlear Implant Programme were recruited in this retrospective cross-sectional study. All paediatric Cochlear Implant (CI) recipients with pre-lingual deafness were included in this retrospective study. The study was conducted from January 2019 until December 2020. The pre-lingual cochlear implant recipients’ data were analysed based on demographics and interval from diagnosis to hearing aid fitting and implantation. The association between the dependent variables with early and late cochlear implantation was compared.
Results: A total of 83 (52%) patients were female. Chinese race constituted most of the patients, which was 90/159 (56%). The majority were from middle-income families (M40); 89 (55.9%). The most common aetiology of Hearing Loss (HL) was idiopathic; 139 (87%), followed by intrauterine infections, which comprised of congenital CMV; 8 (5%) and congenital Rubella; 1 (1%) and nonspecific intrauterine infection 2 (1%). The relationship between the universal neonatal hearing screening and the interval between diagnosis to implantation was significant (p=0.033). Other variables were not significant.
Conclusion: UNHS was a significant factor contributing to early and late implantation. The median age of diagnosis of hearing loss was 18 months (interquartile range; 15); the age of CI was 34 months (interquartile range; 24); the interval from diagnosis to hearing aid was 2 months (interquartile range; 5), and the interval from diagnosis to CI was 16 months (interquartile range; 14).
Keywords: Pre-lingual deafness, Cochlear implantation, Hearing loss
Introduction
Cochlear Implants (CI) have been accepted as an effective treatment for patients with severe-to-profound Sensorineural Hearing Loss (SNHL), whether in prelingual or post-lingual deafness populations [1, 2]. However, the number of cochlear implantation may vary between countries, depending on selection criteria, which are determined mainly by economic restrictions.
The prevalence of mild to profound SNHL is reported to be between 1.1 to 6 per 1000 live birth, with significant implications during the first three years of life in terms of speech and language acquisition subsequently on educational and psychosocial development [3, 4].
Candidates for the CI will undergo preoperative assessment involving clinical, audiological, speech and language, radiological and social criteria. These assessments are paramount to CI surgery to anticipate the outcome, expectations, and complications from the surgery. With the -changing economy and new technology development with emerging evidence-based criteria, the broadest range of individuals can now benefit from CI. In a previous study, among 115 CI candidates with profound sensorineural hearing loss, 52% had prelingual hearing loss. At first consultation, the mean age was 3.8 years, with idiopathic being the most common aetiology [5].
Early exposure of the auditory system to electrical stimulation has been shown to improve the developmental curve for both speech perception and production and language abilities.. However, the optimal age at implantation is still debatable. Nevertheless, children with profound sensorineural hearing loss should receive a CI soon after diagnosis to shorten the period of auditory deprivation [6, 7]. Almost all infants implanted before the age of 12 months achieved language scores in the normal range when tested using Peabody Picture Vocabulary Test at the age of at least six years old [8]. The fact that shorter duration of deafness gives better outcomes is also supported by the previous study, in that greater rates of expressive and receptive language growth were found in children implanted under 12 months of age in comparison to children implanted between 12 and 24 months of age [9]. Thus, supporting the earlier age of implantation yield a better outcome. However, many predictors cause delayed implantation, which needs to be addressed and tackled in the future to promote early CI.
Material and Methods
The retrospective study was conducted at the Hospital Canselor Tuanku Muhriz (HCTM) and the Audiology and Speech Sciences Clinic, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, from January 2019 until December 2020. This study received ethical approval from both the university research ethics committee (UKM PPI/111/8/JEP-021). A total of 159 pre-lingual CI recipients from 2012 until 2020 were included in this study. We reviewed all the clinical records of all recipients of cochlear implants to collect demographic records, the date of the first implant, and the intervention manufacturer and device type.
Pre-lingual deafness is defined by hearing impairment that begins before four years [10]. The analysis was done using Statistical Package for the Social Science (SPSS) version 26. Data were presented as median (interquartile range). Categorical variables were compared using the Chi-square test. All of the enrolled patients agreed to the use of their medical records.
Results
A total of 83 (52%) study participants were female and 76(48%) were males. Chinese race constituted the majority (57%), followed by Malay (34%), Indian (4%), and non- Malaysian (4%). The study participants were categorized based on the household income, which showed that the majority were from middle-income families (M40) i.e., 89 (56%). The number of study populations from the T20 and B40 groups as 38 (24%) and 32 (20%). The majority of the patients were from educated background, whereby most of the participants’ paternal education level was of higher education i.e., 135 (85%). The maternal education level echoed similar findings, whereby the majority of participants were of higher education level; 131 (82%). The study populations mainly live more than 5km away from HCTM; 153 (96%).
Based on the demographic data (Table 1), most study participants, 25% (40), sought the first consultation from medical officers either in primary health clinics or in primary, secondary or tertiary hospitals. This was followed by ENT specialists; 24% (38), audiologists; 22% (35), pediatricians; 21% (33) and private general practitioners and speech therapists; 4% respectively .
| Demographics | No of participants | Percentages (%) |
|---|---|---|
| Gender | ||
| Males | 76 | 48 |
| Females | 83 | 52 |
| Races | ||
| Chinese | 90 | 57 |
| Malays | 53 | 34 |
| Indians | 7 | 4 |
| Non-Malaysians | 7 | 4 |
| Others | 2 | 1 |
| Category of household income* | ||
| B40 | 32 | 20 |
| M40 | 89 | 56 |
| T20 | 38 | 24 |
| Maternal education ** | ||
| Lower education level | 28 | 18 |
| Higher education level | 131 | 82 |
| Paternal education ** | ||
| Lower education level | 24 | 15 |
| Higher education level | 135 | 85 |
| Professionals at first encounter | ||
| Medical officers | 40 | 25 |
| Audiologists | 35 | 22 |
| Speech therapists | 7 | 4 |
| ENT specialists | 38 | 24 |
| Pediatricians’ | 33 | 21 |
| General practitioners | 6 | 4 |
| Distance from hospital (KM) | ||
| <5 KM | 6 | 4 |
| ≥5 KM | 153 | 96 |
**Lower education includes primary and secondary education and upper education level includes pre-university, undergraduate and postgraduate studies.
Table 1: Participants’ demographics information (N=159).
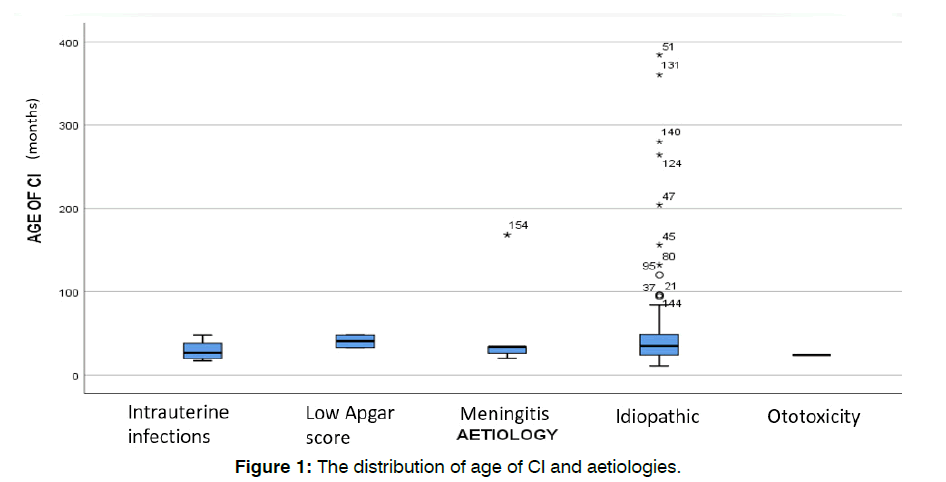
The median age of the first consultation was 17 months (with interquartile range, IQR=16) as compared to the median age of diagnosis, which was 18 months (IQR, 15) (Table 2). The median age of hearing aid fitting was 21 months (IQR,18) and age of implantation was 34 months (IQR, 24). However, the minimum age of implantation was 11 months, whereas the maximum age of implantation was 384 months. The median interval from diagnosis to HA fitting and implantation were 2 months (IQR, 5) and 16 months (IQR, 14), respectively. The most common etiology was idiopathic; 139 (87%), followed by intrauterine infections, which comprised of congenital CMV; 8 (5%) and congenital Rubella; 1 (1%) and nonspecific intrauterine infection 2 (1%). This was followed by meningitis; (4%) and ototoxicity, and low APGAR score representing 1%, respectively. Figure 1 showed that the distribution of age of CI was wide-ranging in those with the idiopathic cause of hearing loss.
| Distribution of age and interval | Age (months) |
|---|---|
| Median (IQR) | |
| Age of the first consultation | 17 (16) |
| Age of diagnosis | 18 (15) |
| Age of hearing aid fitting | 21 (18) |
| Age of implantation | 34 (24) |
| Interval from diagnosis to HA fitting |
2 (5) |
| Interval from diagnosis to implantation |
16 (14) |
Table 2: The median age and interval of the study participants.
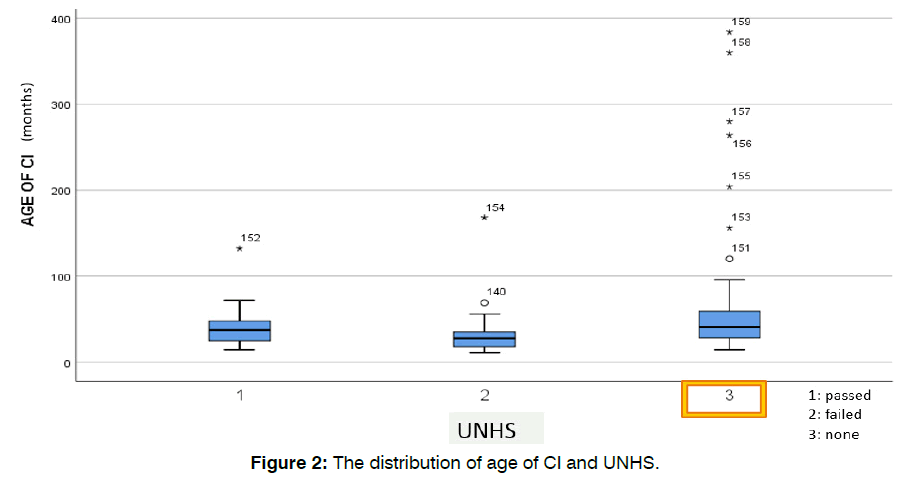
HCTM has screened a total of 41,887 neonates through the UNHS program from 2012 until 2020. A sum of 81 patients (51%) from 159 subjects was screened. Based on the box-plot (Figure 2), the distribution of age of CI varied in those who did not undergo UNHS. Compared with other dependent variables, the relationship between the dependent variable with early and late cochlear implantation was analysed using the Chi-Square test. The relationship between UNHS with the interval from diagnosis to implantation was significant (Table 3).
| Implantation | UNHS | ||
|---|---|---|---|
| Passed | Failed | p-value | |
| Early | 14 (48%) | 15 (52%) | |
| Late | 13 (25%) | 39 (75%) | 0.033 |
Table 3: The association between Universal Neonatal Hearing Screening (UNHS) with the interval from diagnosis to implantation.
Discussion
Cochlear implantation has become an accepted treatment paradigm for individuals with bilateral severe to profound SNHL [11]. Based on recent data, approximately 324,200 people worldwide have received cochlear implants as of December 2012, and in Malaysia, the first cochlear implant was performed in 1995 at Universiti Kebangsaan Malaysia Cochlear Implant programme [12].
It has been well recognized that early implantation is highly desirable to maximize the child’s access to speech sounds during periods of maximum neural plasticity that in turn should lead to the development of more age-appropriate speech-language skills [5, 13]. Universal hearing screening of newborns is the cornerstone of early identification and diagnosis of hearing loss in newborns. Hearing loss in pediatric age can remarkably impact language acquisition and educational and socialemotional development, which presents a significant burden to the quality of life in the future4. The Joint Committee on Infant Hearing guidelines (2019) suggested that a newborn needs to have a diagnosis of hearing loss confirmed by 3 months old. and receive early intervention before 6 months old [14].
Our data showed there was a significant relationship between UNHS and cochlear implantation. The importance of UNHS and the relationship with age of implantation were mentioned in previous studies [15-17]. Children born after legally mandated UNHS had significantly younger ages at diagnosis and implantation. However, a younger age at diagnosis of SNHL was not achieved in children who had passed UNHS or who were not screened17. A recent study revealed that children referred via UNHS were implanted at a younger age than those referred to pre-UNHS, which has a positive impact on children’s functional outcomes [18].
The present study showed that the median age of the first consultation was 17 months (IQR, 16) as compared to the median age of diagnosis which was 18 months (IQR, 15). The median age of hearing aid fitting and age of implantation were 21 months (IQR, 18) and 34 months (IQR, 24), respectively. In a relatable local study done by Goh et al. (2018), among the pediatrics cochlear implant recipients, the mean age of diagnosis was 26 months, while the mean age of implantation was 39.8 months [19]. Early and late implantation were defined by the interval from diagnosis to implantation of <12 months and ≥ 12 months, respectively, whereby most were late implantation; 107 (67%) [13].
Chinese race constituted most of the pre-lingual cochlear implant recipients, followed by Malays, Indians, and Non Malaysians (Burmese, Vietnamese and Indonesian). Other races comprised of Native Borneo were less than 1%. The relationship between race and cochlear implantation was not significant. A study reported that race or ethnicity was not associated with hearing thresholds after stratification by skin color, which indicates that skin color is independently associated with hearing loss [20]. In a study in the United States by Stern et al. [21], White and Asian children were implanted at higher rates than Hispanic and black children, which can be related to socioeconomic status [21]. Malaysia is a multiracial country that is also rich in culture and tradition. However, we have no local data yet to associate race with early and late implantation.
The most common etiology was idiopathic; 139 (87%), followed by intrauterine infection and meningitis. Our present study reported similar findings based on the previous research by Barbosa et al5. 33% of congenital HL was idiopathic. In several studies carried out in the United States from 1966 to 2007, in 56% of cases, the etiology of hearing loss was unknown and among the known aetiologies, genetic causes were the most prevalent [22, 23].
Access to healthcare is a complex issue that is determined by multiple factors that intermingled with each other. The risk of non-compliance is higher in families with greater travel distances, low levels of parental education, low socioeconomic status, and public insurance [24]. A systematic review discovered that educational disparities, travel distance, work constraints, unfavorable attitudes, and competing healthcare needs were the primary reasons for high loss to follow up in UNHS [25]. Kothari et al. (2015) attributed early and late implantation to factors such as distance to the tertiary center, comorbidities that required more urgent interventions, and financial support. The parents’ illiteracy may play a role in delayed investigations and delayed in decision-making [26]. However, there was no association between parental education and early and late implantation. The proposed distance between household and medical facilities varied with each country. The majority of 88.5 % of the population lives within 5 km of a health facility (Merican, 2007) [27, 28]. HCTM of the CI recipients live more than 5km from UKMMC, which is a tertiary center. The association between the distance from hospital with implantation was not statistically significant. A similar finding was reported by Al Shawi et al (2020) [29].
Delay to follow-up for children with hearing loss in private health care can be related to socioeconomic status, which appears less likely in a publicly funded government hospital [26]. In 2019, Malaysia’s mean household income was RM7, 901 while Malaysia’s median income recorded at RM5, 873. The mean household income in Malaysia is further categorized into B40; less than RM4, 850, M40; between RM4, 850 and RM10, 959 and T20; RM10, 960 [27]. According to our present study, 56% (89) falls into the M40 category. There was no significant association between the interval from diagnosis and implantation and the household income. The majority of the CI recipients were financially supported by the non-governmental association, corporate organizations and governmentlinked funds.
Conclusion
UNHS was a significant factor contributing to early and late implantation. The median age of diagnosis was 18 months (IQR:15); the age of CI was 34 months (IQR, 24); the interval from diagnosis to HA was two months (IQR, 5), and the interval from diagnosis to CI was 16 months (IQR, 14). This study has highlighted the importance of UNHS in the university hospital.
References
- Wooi Teoh S, Pisoni DB, Miyamoto RT. Cochlear implantation in adults with prelingual deafness. Part I. Clinical results. Laryngoscope. 2004;114(9):1536-40.
- Im GJ, Ahn JH, Lee JH, Do Han K, Lee SH, Kim JS, et al. Prevalence of severe-profound hearing loss in South Korea: a nationwide population-based study to analyse a 10-year trend (2006–2015). Sci Rep. 2018;8(1):9940.
- Abdullah A, Hazim MY, Almyzan A, Jamilah AG, Roslin S, Ann MT, et al. Newborn hearing screening: experience in a Malaysian hospital. Singapore Med J. 2006;47(1):60.
- Vincenti V, Bacciu A, Guida M, Marra F, Bertoldi B, Bacciu S, et al. Pediatric cochlear implantation: an update. Ital J Pediatr. 2014;40(1):1-7.
- Barbosa MH, Felix F, Ribeiro MG, Tomita S, Pinheiro C, Baptista MM. Profile of patients assessed for cochlear implants. Braz J Otorhinolaryngol. 2014;80:305-10.
- Bruijnzeel H, Bezdjian A, Lesinski-Schiedat A, Illg A, Tzifa K, Monteiro L, et al. Evaluation of pediatric cochlear implant care throughout Europe: Is European pediatric cochlear implant care performed according to guidelines?. Cochlear Implants Int. 2017;18(6):287-96.
- Niparko JK, Tobey EA, Thal DJ, Eisenberg LS, Wang NY, Quittner AL, et al. Spoken language development in children following cochlear implantation. JAMA. 2010;303(15):1498-506.
- Miyamoto RT, Colson B, Henning S, Pisoni D. Cochlear implantation in infants below 12 months of age. World J Otorhinolaryngol Head Neck Surg. 2017;3(04):214-8.
- Dettman SJ, Pinder D, Briggs RJ, Dowell RC, Leigh JR. Communication development in children who receive the cochlear implant younger than 12 months: risks versus benefits. Ear Hear. 2007;28(2):11S-8S.
- Clark GM, Blamey PJ, Brown AM, Gusby PA, Dowell RC, Franz BK, et al. The University of Melbourne--nucleus multi-electrode cochlear implant. Adv Otorhinolaryngol. 1987;38:V-IX.
- Shakrawal N, Sonkhya N, Agarwal S, Grover M. The effect of age at cochlear implantation on speech and auditory performances in prelingually deaf children. Indian J Otolaryngol Head Neck Surg. 2020:1-0.
- Devesahayam PR, Orlhns M, Hashim SS, Orlhns M, Salahuddin Z, Kamalden TM, et al. Surgical and functional outcomes of cochlear implantation in post-lingual and cross-over patients: first 5-year review of the National Ministry of Health Malaysia cochlear implant programme. Med J Malaysia. 2018;73(6):393.
- Fitzpatrick EM, Ham J, Whittingham J. Pediatric cochlear implantation: why do children receive implants late?. Ear Hear. 2015;36(6):688.
- Yusoff YM, Umat C, Mukari SZ. Profiling the national cochlear implant recipients for prelingual, hearing-impaired children in Malaysia. Malaysian J Health Sci. 2017;15(2):153-62.
- Li W, Dai C, Li H, Chen B, Jiang Y. Factors impacting early cochlear implantation in Chinese children. Eur Arch Otorhinolaryngol. 2016;273:87-92.
- Mohr PE, Feldman JJ, Dunbar JL, McConkey-Robbins A, Niparko JK, Rittenhouse RK, et al. The societal costs of severe to profound hearing loss in the United States. Int J Technol Assess Health Care. 2000;16(04):1120-35.
- Young NM, Reilly BK, Burke L. Limitations of universal newborn hearing screening in early identification of pediatric cochlear implant candidates. Arch Otolaryngol Head Neck Surg. 2011;137(3):230-4.
- Gabriel MM, Geyer L, McHugh C, Thapa J, Glynn F, Walshe P, et al. Impact of Universal Newborn Hearing Screening on cochlear implanted children in Ireland. Int J Pediatr Otorhinolaryngol. 2020;133:109975.
- Goh BS, Fadzilah N, Abdullah A, Othman BF, Umat C. Long-term outcomes of universiti Kebangsaan Malaysia cochlear implant program among pediatric implantees. Int J Pediatr Otorhinolaryngol. 2018;105:27-32.
- Lin FR, Maas P, Chien W, Carey JP, Ferrucci L, Thorpe R. Association of skin color, race/ethnicity, and hearing loss among adults in the USA. J Assoc Res Otolaryngol. 2012;13:109-17.
- Stern RE, Yueh B, Lewis C, Norton S, Sie KC. Recent epidemiology of pediatric cochlear implantation in the United States: disparity among children of different ethnicity and socioeconomic status. Laryngoscope. 2005;115(1):125-31.
- Miyagawa M, Nishio SY, Usami SI. A comprehensive study on the etiology of patients receiving cochlear implantation with special emphasis on genetic epidemiology. Otol Neurotol. 2016;37(2):e126.
- Mehra S, Eavey RD, Keamy Jr DG. The epidemiology of hearing impairment in the United States: Newborns, children, and adolescents. Otolaryngol Head Neck Surg. 2009;140(4):461-72.
- Bush ML, Kaufman MR, McNulty BN. Disparities in access to pediatric hearing healthcare. Curr Opin Otolaryngol Head Neck Surg. 2017;25(5):359.
- Ravi R, Gunjawate DR, Yerraguntla K, Lewis LE, Driscoll C, Rajashekhar B. Follow-up in newborn hearing screening–A systematic review. Int J Pediatr Otorhinolaryngol. 2016;90:29-36.
- Kothari S, Keshree NK, Bhatnagar S. Pediatric cochlear implantation—why the delay. Indian J Otolaryngol Head Neck Surg. 2015;67:165-9.
- Malaysia DO. Department of statistics Malaysia official portal. The source of Malaysia's official statistics. 2019.
- Shariff SS, Moin NH, Omar M. Planning of public healthcare facility using a location allocation modelling: A case study. In AIP Conference Proceedings. 2014;1613:282-296.
- Al-Shawi YA, Alrawaf FK, Al-Gazlan NS, Al-Qahtani MM, Almuhawas FA. Relationship between proximity to a cochlear implant center and early presentation in children with congenital hearing loss. Saudi Med J. 2020;41(3):314.
1Department of Otorhinolaryngology Head and Neck Surgery, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
2Centre for Rehabilitation and Special Needs Studies, Faculty of Health Sciences, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
3Epidemiology and Statistics, Department Of Community Health, Universti Kebangsaan Malaysia, Kuala Lumpur, Malaysia
Send correspondence to:
Dr Asma Abdullah
Department of Otorhinolaryngology Head and Neck Surgery, Faculty of Medicine, Universiti Kebangsaan Malaysia, Kuala Lumpur, Malaysia, Email: asmappukm@gmail.com
Tel no: 391456599
Paper submitted on February 24, 2023; and Accepted on March 20, 2023
Citation: Mansor WNW, Abdullah A, See GB, Umat C, Shah SA. Factors Affecting Early and Late Cochlear Implantation. Int Tinnitus J. 2023;27(1):34-39.